U of T Scientists Cause Ripple in Innate Immune Research

Professor James Carlyle was unconvinced at first. His doctoral student, Oscar Aguilar, had just brought him lab data showing how ‘natural killer’ or NK cells, a key part of the immune system’s defense against pathogens and cancer, are targeted by a common virus.
“I’d been working on this for so long — my scientific lifetime — that I was a bit skeptical at first,” says Carlyle, a professor in the Department of Immunology at the University of Toronto and a senior scientist at Sunnybrook Research Institute.
“I was pretty hard on him, asking for more detailed and better data — optimal assays, higher-affinity alleles [genetic variants], more striking phenotypes.” He even had Aguilar re-make the reporter cells, which the lab uses to track protein interactions, to be sure they weren’t contaminated.
Finally, both were convinced their results were accurate. A viral decoy protein called m12, derived from mouse cytomegalovirus, or MCMV — a virus similar to human CMV that can cause serious health problems — binds to a key family of NK cell receptors and inhibits NK cell function.
“The first NK cell receptor was discovered 40 years ago, but it’s been an orphan receptor ever since,” says Carlyle. “A number of other NK cell receptors and their ligands [binding partners] have been identified, but the physiological partner for NK1.1 — the prototypical NK receptor — has been a mystery.”
The journal Cell published the results online last month.
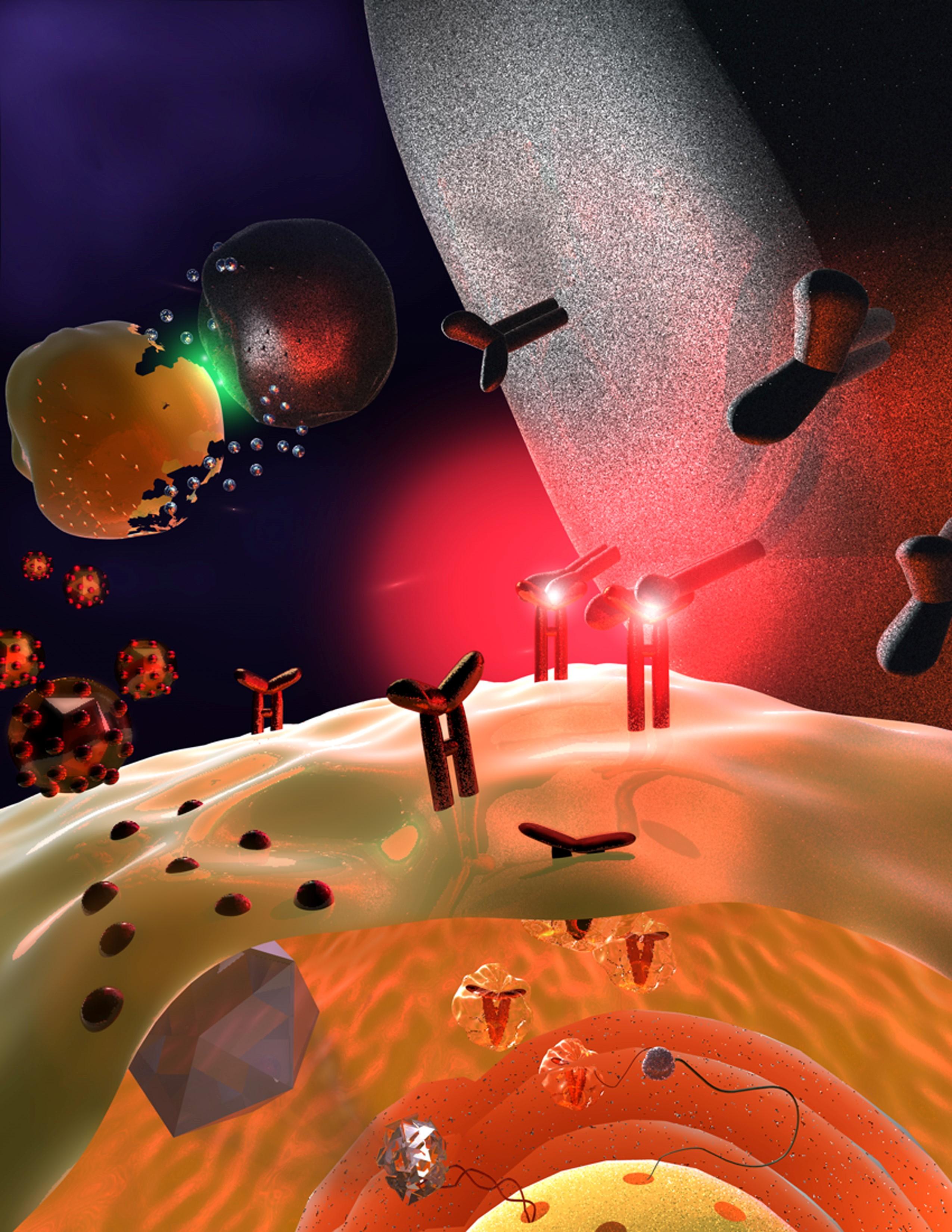 The findings provide a deeper understanding of the evolutionary interplay between viruses and NK cells, white blood cells that act as sentinels for the immune system. Aguilar likens that interplay to an evolutionary arms race, with each side gaining momentum at various points over millennia.
The findings provide a deeper understanding of the evolutionary interplay between viruses and NK cells, white blood cells that act as sentinels for the immune system. Aguilar likens that interplay to an evolutionary arms race, with each side gaining momentum at various points over millennia.
Right now, the virus has the upper hand. It tricks the immune system by making viral cells appear as healthy cells, in part by binding to an inhibitory receptor that prevents NK cells from attacking; however, in some mice it also binds to a stimulatory NK cell receptor. Critically, the virus wins when inhibition takes place, but the host wins when activation occurs.
If a similar process is at work in human cells — and Carlyle’s lab has some early evidence that it may be — the results could provide the basis for new treatments for human CMV. Most people carry the virus without knowing it, but it can cause major organ damage and even death in newborns and people with weakened immune systems.
One potential treatment option is passive immunotherapy, in which elements of the immune system such as antibodies are engineered in the lab and given to patients; another is a vaccine, which stimulates the body’s own immune response.
Aguilar plans to be part of experiments in Carlyle’s lab with human NK cell receptors and human CMV over the next few months. But since Cell published his research, he has fielded aggressive offers for postdoctoral positions at high-profile labs in the U.S. and other countries. “It makes me a bit nervous, but it’s also really exciting,” Aguilar says. He successfully defended his doctoral thesis in the Department of Immunology last week.
For Carlyle, the research culminates 22 years of work on NK cells, and more than a decade of international collaborations on the NK cell receptor family. In 2005, he began work with Professor Andrew Makrigiannis, currently head of microbiology and immunology at Dalhousie University, on the genetics of the NK cell receptor family. That led to their identification of host genetic variations called allelic polymorphisms in mice and rats.
The Carlyle lab also worked with Dr. Munir Rahim of the Makrigiannis lab, then at the University of Ottawa, on MCMV interactions in knockout mice (animals lacking specific genes). They later worked with Professor Stipan Jonjić at the University of Rijeka in Croatia, whose lab genetically manipulates the MCMV genome to study immunoevasins that trick the immune system. They created mutant viruses lacking specific genes and then homed in on the m12 gene.
The final piece of the puzzle came in 2015, when Carlyle and Aguilar went to an international Society for Natural Immunity NK meeting in Montebello, Quebec. There they met Dr. Richard Berry, a structural immunologist from the lab of Professor Jamie Rossjohn at Monash University in Australia. They sent him genetic data and material for the NK cell receptors, ligands, and alleles from different mouse and virus strains, and within a year he had produced a crystal structure that showed atomic-level detail on how m12 binds to the NK cell receptor family.
The crystal was one of the few parts of the project that went smoothly. “Pretty much everything was a challenge, all the way along,” says Aguilar. He had limited access to knockout mice, the parental viral strain was a challenge, and the MCMV genome has over 200 genes, so it took him months to cut out a small fraction of it and test about 30 independent genes, of which m12 signified the Eureka moment.
 “The project did start slowly,” agrees Carlyle, who notes that Aguilar began his work in the lab on a different project, but multitasked on the NK cell ligand research and switched to it when he got an unequivocal positive result.
“The project did start slowly,” agrees Carlyle, who notes that Aguilar began his work in the lab on a different project, but multitasked on the NK cell ligand research and switched to it when he got an unequivocal positive result.
From that point on, it was a question of perseverance. “The main thing I learned is that doing a doctoral degree is a struggle with yourself,” says Aguilar. “I had so many failed experiments, but if I gave up I would have missed out. Sometimes you have to follow your intuition, and mine was telling me, ‘keep going.’”
Funding for the study was provided by the Burroughs Wellcome Fund, Canadian Institutes of Health Research, Natural Sciences and Engineering Research Council of Canada, and the Ontario Ministry of Research, Innovation and Science, with support from other international agencies.
Read the media release on this study from Sunnybrook Research Institute.
News


